Lichen butyrolactone derivatives disrupt oral bacterial membrane
Résumé
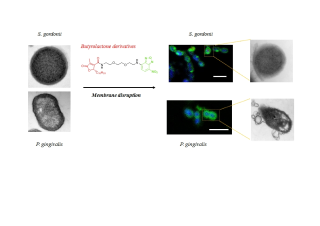
We have previously demonstrated that out of the butyrolactones series synthesized based on the natural lichen metabolite lichesterinic acid, compound (B-13) was the most effective against oral bacteria. However, its antibacterial mechanism is still unknown. In this study, we have investigated its bacterial localization by synthesizing a fluorescently labeled B-13 with NBD while maintaining its antibacterial activity. We showed that this compound binds to Streptococcus gordonii cell surface, as demonstrated by HPLC analysis. By adhering to cell surface, B-13 induced cell wall disruption leading to the release of bacterial constituents and consequently, the death of S. gordonii, a Gram-positive bacterium. A Gram-negative counterpart, Porphyromanas gingivalis, showed also cracked and ruptured cells in the presence of B-13. Besides, we also demonstrated that the analog of B-13, B-12, has also induced disruption of P. gingivalis and S. gordonii. This study revealed that butyrolactones can be considered as potent antibacterial compounds against oral pathogens causing medical complications.
Fichier principal
 Sweidan-2019-Lichen butyrolactone derivatives.pdf (5.11 Mo)
Télécharger le fichier
Sweidan-2019-Lichen butyrolactone derivatives.pdf (5.11 Mo)
Télécharger le fichier
Origine : Fichiers produits par l'(les) auteur(s)
Loading...

